“We want to make life easier for our customers when it comes to procuring their dropper bottles and, by treating our products with gamma radiation, we are adding in an important work step before filling,” says Niels Düring, Global Executive Vice President Plastic Packaging. “To help us in this, we are relying on selected, recognized, and certified partners.” The chosen partner companies have the requisite certification in accordance with ISO standards 11137, 11737, and 13004.
Benefits
The benefits of this service speak for themselves: Gerresheimer is assuming responsibility for handling this work step from start to finish, including transport to the irradiation company, monitoring and inspecting its work, and delivering to the location specified by the customer. The inspection processes also involve physically and chemically testing the product characteristics after irradiation. Gerresheimer regulates the validation and revalidation of the entire process, helping to reduce costs for the customer.
Cleanroom production
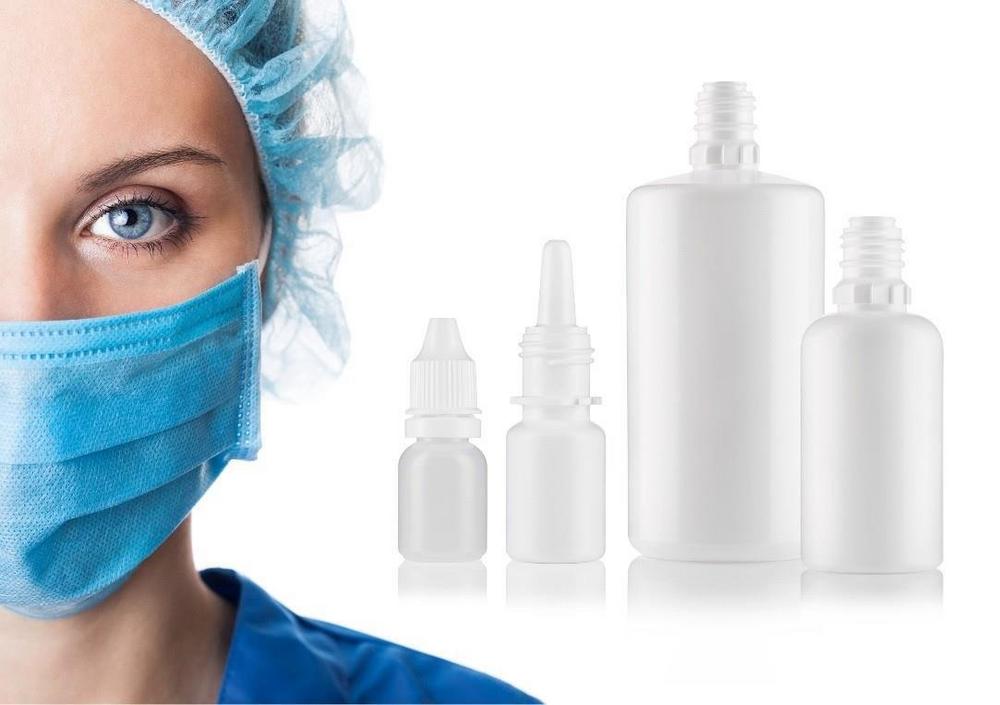
Gerresheimer’s range of ophthalmology and rhinology products encompasses bottles and dropper inserts made from lowdensity polyethylene (LDPE) with pump systems to match. Irradiation also ensures the products are germfree. As a specialist in plastic packaging for the pharmaceutical industry, Gerresheimer offers a wide range of innovative packaging solutions for solid, liquid, and opthalmological products. All of the company’s primary packaging for the pharmaceutical industry is produced in ISO class 7 and class 8 cleanrooms at its plants in Vaerloese (Denmark) and Bolesławiec (Poland).
Low germ level thanks to ISO-standard irradiation
The population of viable microorganisms on the surface of a product and/or packaging is called the bioburden. The bioburden is determined in accordance with ISO 11737. Raw materials, components, packaging, and medical products are all investigated in order to gather information about the germ composition and level of germ contamination before treatment. A stable bioburden guarantees a successful irradiation process.
Gerresheimer will be at booth 4C30 of CPhI Worldwide in Madrid (IFEMA) from October 9 to 11 and from October 14 to 17 at booth W-964 of Pack Expo in Chicago (McCormick Place)..
Further info
Gerresheimer is a leading global partner to the pharma and healthcare industry. With specialty glass and plastic products, the Company contributes to health and wellbeing. Gerresheimer operates worldwide and its approximately 10,000 employees manufacture products in local markets, close to its customers. With our plants in Europe, North America, South America, and Asia, we generate revenues of approximately EUR 1.4 billion. The comprehensive product portfolio includes pharmaceutical packaging and products for the safe, simple administration of medicines: Insulin pens, inhalers, prefillable syringes, injection vials, ampoules, bottles, and containers for liquid and solid medicines with closure and safety systems as well as packaging for the cosmetics industry.
Gerresheimer AG
Klaus-Bungert-Straße 4
40468 Düsseldorf
Telefon: +49 (211) 6181-00
Telefax: +49 (211) 6181-295
http://www.gerresheimer.de
Senior Manager Corporate Communication
Telefon: +49 (211) 6181-246
Fax: +49 (211) 6181-28246
E-Mail: m.stolzenwald@gerresheimer.com
Group Senior Director Communication & Marketing
Telefon: +49 (211) 6181-250
Fax: +49 (211) 6181-241
E-Mail: J.Kuerten@gerresheimer.com